Hydrometallurgical Process for Pure Metal Extraction
Metals are the unsung heroes of our modern world, forming the backbone of countless technologies that we rely on every day. From the tiny chips in our smartphones to the sturdy beams holding up our buildings and the energy systems that keep our world running, metals—especially in their purest form—are more essential than ever. For years, extracting metals meant using extreme heat through pyrometallurgy. But now, a growing and innovative approach called hydrometallurgy is changing the game—offering a potentially cleaner and more sustainable way to obtain these crucial materials.
Instead of relying on high temperatures, hydrometallurgy, in essence, is the science of using aqueous solutions to extract and purify metals from various sources, including ores freshly mined from the earth, concentrates that have undergone initial processing, and even the increasingly important realm of recyclable materials like electronic waste. Often called “wet metallurgy,” this process is known for yielding exceptionally pure metals and is also thought to be a more environmentally friendly form of extraction.
The Process of Hydrometallurgy
Under the broad category of extractive metallurgy, this technique recovers metals from their ores, concentrates, and recycled or residual materials using aqueous solutions. This process is a cornerstone of extractive metallurgy, which is the process of removing metals from both recycled materials and their natural sources, such as ores and concentrates. To extract these precious metals, water-based treatments are used.
While the large-scale prominence of hydrometallurgy came in the 20th century, its origins can be traced back much further. Although metallurgy emerged in the 20th century, its roots go far deeper. Water-based solutions have been used for metal extraction since the 16th and 17th centuries, according to historical data. But the true force behind the development of these methods in the late 19th and early 20th centuries was the desire to efficiently extract gold from inferior ores. Today, hydrometallurgical processes are employed to recover over 70 metallic elements, demonstrating its versatility and importance in modern materials science and engineering.
Hydrometallurgy vs. Pyrometallurgy:
Comparing Extraction PhilosophiesTo truly appreciate the role of hydrometallurgy, it’s essential to understand its relationship with pyrometallurgy, the other major branch of extractive metallurgy. Pyrometallurgy relies on high temperatures to drive chemical reactions that separate metals from their ores through processes like roasting, smelting, and refining. While pyrometallurgy remains crucial for certain metals and ore types, hydrometallurgy offers distinct advantages in several areas.
One key difference lies in energy consumption. Hydrometallurgy often requires lower overall energy input, especially when processing significant financial savings and a less carbon footprint may result from this. When it comes to the environment, hydrometallurgy typically emits fewer air pollutants than pyrometallurgy, which uses high temperatures and can release pollutants like sulphur dioxide. However, it’s important to note that hydrometallurgical processes can generate wastewater containing dissolved salts and reagents, requiring careful treatment and disposal.
In terms of versatility, hydrometallurgy often excels at treating complex ores and is particularly well-suited for selectively extracting specific metals from mixtures, such as those found in electronic waste. Pyrometallurgy, on the other hand, is often more effective for ores with high metal concentrations and stable mineral compositions. Ultimately, the choice between these two metallurgical approaches depends on a variety of factors, including the specific metal being extracted, the nature of the ore, economic considerations, and environmental regulations. In many modern applications, especially in the rapidly growing field of battery and e-waste recycling, hydrometallurgy is increasingly recognized as the more sustainable and resource-efficient option.
| Feature | Hydrometallurgy | Pyrometallurgy |
| Principle | Uses aqueous solutions for dissolution and recovery | Uses high temperatures for smelting and refining |
| Energy Consumption | Generally lower, especially for low-grade ores | Generally higher, requiring significant fuel sources |
| Environmental Impact | Lower air emissions, potential for wastewater issues | Higher air emissions, less wastewater but potential for toxic byproducts in slag |
| Ore Suitability | Well-suited for low-grade, complex ores, and recycled materials | Best for high metal content ores with stable mineral compositions |
| Metal Recovery | High recovery rates for many metals, allows for selective extraction | Effective for certain metals, can be less selective |
The Hydrometallurgical Process: A Step-by-Step Guide
The extraction of pure metals through hydrometallurgy is a carefully controlled process involving several key stages:
Leaching: Dissolving the Treasure
The initial and often most crucial step is the leaching process in metallurgy. This involves contacting the metal-bearing material with an aqueous solution, known as a lixiviant or leaching agent, which selectively dissolves the desired metal while leaving behind the bulk of the unwanted material. The choice of leaching method and agent is highly dependent on the specific metal and the characteristics of the ore or concentrate.
Various leaching techniques are employed depending on the scale and nature of the operation. Heap leaching is a cost-effective method for processing large volumes of low-grade ores, where the crushed ore is piled into heaps and the leaching solution is sprayed over the top, percolating down to dissolve the metal. For finer materials or higher-grade ores, vat leaching or tank leaching (also known as agitation leaching) might be used, where the material is immersed or slurried in the leaching solution within large tanks, often with mechanical agitation to enhance the dissolution rate. In some cases, for deeply buried deposits, in-situ leaching (or solution mining) is employed, where the leaching solution is pumped directly into the ore body underground. For more resistant materials, autoclave leaching (or pressure leaching) can be used, where the leaching is carried out in closed reactors under high temperature and pressure.
The leaching agents themselves are diverse, ranging from acids like sulfuric acid (commonly used for copper and uranium) and hydrochloric acid, to bases like sodium hydroxide, and complexing agents like cyanide (essential for gold and silver extraction) and ammonia (used for certain copper and nickel ores). Even microorganisms can play a role in bioleaching, assisting in the dissolution of metals.
Solution Purification: Separating the Wheat from the Chaff
The solution obtained after leaching, known as the pregnant leach solution (PLS), contains the dissolved metal of interest but also a variety of impurities. Solution purification is a crucial step to selectively remove these unwanted elements, ensuring a high-purity final metal product. Several techniques are employed for this purpose:
- Cementation: As discussed later, this process can also be used to remove unwanted metal ions from the solution.
- Precipitation: Specific chemical reagents are added to the solution to selectively precipitate out impurities as insoluble solids, which are then removed by filtration or settling. For instance, copper sulfide can be precipitated to purify nickel leachates.
- Solvent Extraction (SX): This method involves using an organic solvent that is immiscible with water to selectively extract the desired metal ion from the aqueous solution, leaving the impurities behind.
- Ion Exchange (IX): Specialized resins are used to selectively exchange ions with the solution, either removing impurities or concentrating the desired metal.
- Filtration: Used to remove any solid particles from the solution.
- Carbon Adsorption: Activated carbon can adsorb certain impurities or even the target metal in specific applications.
Metal Recovery: Bringing the Metal Back to Solid Form
Once the solution is purified, the next crucial step is metal recovery, where the dissolved metal ions are converted back into their pure metallic form. Common methods include:
- Electrowinning (EW): An electrochemical process where an electric current is passed through the purified solution, causing the metal ions to be reduced and deposited as pure metal onto a cathode. This is a primary method for copper recovery.
- Precipitation: Adding specific reagents can cause the dissolved metal to precipitate out of the solution as a solid.
- Gaseous Reduction: Treating the purified solution with reducing gases like hydrogen can directly produce metal powder.
Refining: Achieving the Highest Purity
In some cases, particularly when ultra-high purity metals are required, a final refining step might be necessary. This can involve techniques like distillation, zone refining, or further electrolytic refining to eliminate any remaining trace impurities.
A Closer Look at the Cementation Process in Hydrometallurgy
The cementation process in hydrometallurgy is a specific and historically significant metal recovery technique. It’s a type of precipitation that occurs through a redox reaction at a solid metallic interface. In this process, a more electropositive metal (higher on the galvanic series) is added to a solution containing ions of a less electropositive metal. The more electropositive metal dissolves (oxidizes), releasing electrons that reduce the less electropositive metal ions in the solution, causing them to precipitate out as solid metal.
A classic application of this is the recovery of copper from acidic leach solutions using scrap iron. The iron (Fe) is more reactive than copper (Cu), and the reaction proceeds as: Cu<sup>2+</sup>(aq) + Fe(s) → Cu(s) + Fe<sup>2+</sup>(aq). The copper precipitates as a solid on the surface of the iron.
The cementation process in hydrometallurgy is also famously used in the Merrill-Crowe process for recovering gold and silver from cyanide leach solutions using zinc dust. Zinc is more electropositive than both gold and silver, leading to their precipitation. This method has been employed for over a century and accounts for a significant portion of global gold production. Cementation can also be used for purifying solutions by removing unwanted metal ions.
Examples in Action: Hydrometallurgy Unveiled
The versatility and effectiveness of hydrometallurgical processes are best illustrated through specific examples, such as the extraction of gold and copper.
The Hydrometallurgical Extraction of Gold
The extraction of gold by hydrometallurgy is a well-established and widely used process, particularly for low-grade gold ores. The most common technique is gold cyanidation, where the crushed ore is leached with a dilute solution of sodium or potassium cyanide in the presence of air. This process forms a water-soluble gold cyanide complex, [Au(CN)<sub>2</sub>]<sup>-</sup>.
The gold is then typically recovered from this solution using the Merrill-Crowe process, which employs zinc cementation. Zinc dust is added to the gold-bearing cyanide solution, and because zinc is more reactive than gold, it displaces the gold ions, causing metallic gold to precipitate out. The chemical reaction is: 2[Au(CN)<sub>2</sub>]<sup>-</sup>(aq) + Zn(s) → [Zn(CN)<sub>4</sub>]<sup>2-</sup>(aq) + 2Au(s). This process allows for efficient recovery of gold from even low concentrations in the leach solution.
How is Copper Extracted by Hydrometallurgy?
Copper can be extracted by hydrometallurgy through various methods, particularly from oxide ores and low-grade sulfide ores. A common and effective approach involves leaching with dilute sulfuric acid, often using heap leaching for large-scale operations with low-grade ores. The sulfuric acid dissolves the copper, forming a copper sulfate solution.
This copper-rich solution then typically undergoes solvent extraction to concentrate and purify the copper. An organic extractant selectively removes the copper ions from the aqueous solution. The copper is then stripped from the organic phase using a strong acid solution, resulting in a purified and concentrated copper sulfate electrolyte. Finally, the copper is recovered in its pure metallic form through electrowinning, where copper ions in the electrolyte are reduced and deposited as solid copper on cathodes. This leaching-solvent extraction-electrowinning (L-SX-EW) process is a cornerstone of modern copper production.
A Matter of Chemistry: Copper vs. Zinc in Hydrometallurgy
The question of copper can be extracted by hydrometallurgy but not zinc why is a common point of inquiry. While it’s not entirely accurate to say that zinc cannot be extracted by hydrometallurgy, the direct leaching and electrowinning process that works so effectively for copper faces more challenges with zinc.
One primary reason relates to the electrochemical properties of the two metals. Copper is less reactive (more noble) than zinc. In electrowinning, metals with higher reduction potentials are more easily deposited onto the cathode. Copper has a significantly higher reduction potential compared to zinc. This means that during electrolysis, copper ions will preferentially be reduced to metallic copper, while zinc requires more energy and specific conditions for efficient electrodeposition.
Furthermore, zinc is more reactive with acids than copper, which can lead to unwanted side reactions during leaching, such as the evolution of hydrogen gas, potentially reducing the efficiency and safety of the process. While hydrometallurgical routes for zinc extraction do exist and are important, particularly for processing certain types of ores and recovering zinc from secondary sources like brass, primary zinc production has traditionally relied more heavily on pyrometallurgical methods due to their efficiency in handling the common zinc sulfide ores.
Hydrometallurgy: A Champion of Metal Recycling
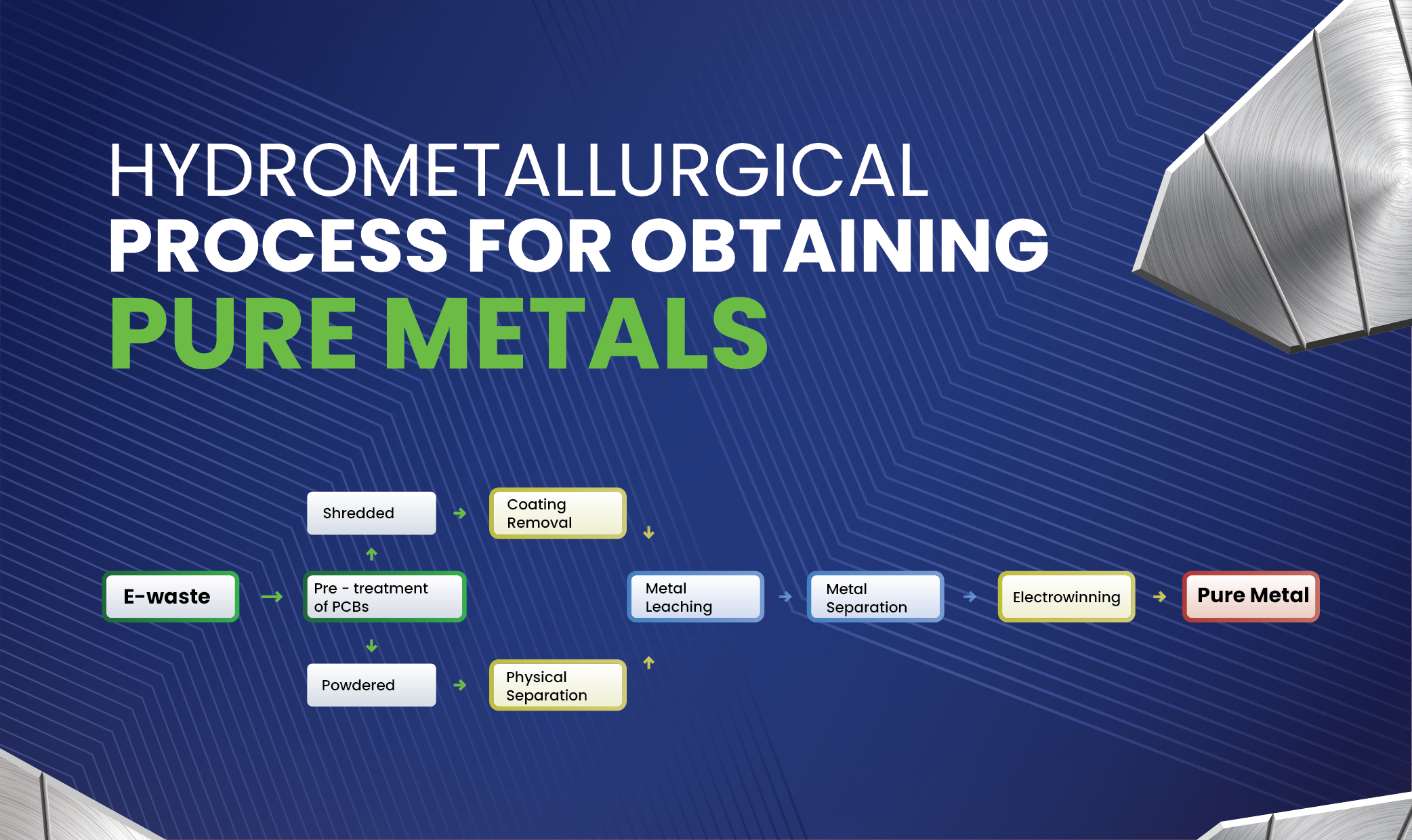
Beyond primary metal extraction, hydrometallurgy recycling is playing an increasingly critical role in recovering valuable metals from various waste streams, especially electronic waste (e-waste) and spent lithium-ion batteries. These complex materials contain a treasure trove of metals, including gold, silver, copper, nickel, cobalt, lithium, and manganese.
Compared to pyrometallurgical recycling, hydrometallurgical methods often offer significant environmental benefits, including lower energy consumption and reduced emissions of greenhouse gases and other pollutants. The process typically involves leaching the shredded waste material with appropriate aqueous solutions to dissolve the metals, followed by a series of purification and recovery steps, such as solvent extraction, precipitation, and electrowinning, to selectively separate and obtain the individual metals. This approach allows for the recovery of a wider range of metals with high purity, contributing to a more circular economy and reducing the reliance on primary mining.
Conclusion: Embracing the Power of Aqueous Solutions
In conclusion, hydrometallurgical processes represent a sophisticated and increasingly important approach to extracting pure metals from a variety of sources. From the selective dissolution of valuable metals through leaching to the precise removal of impurities and the final recovery of high-purity metals, hydrometallurgy offers a versatile and often more sustainable pathway compared to traditional high-temperature methods. Its critical role in both primary metal production and the burgeoning field of metal recycling positions it as a cornerstone of modern metallurgy, essential for meeting the growing global demand for pure metals in an environmentally responsible manner.
Frequently Asked Questions (FAQs)
What is Hydrometallurgy?
Hydrometallurgy is a branch of extractive metallurgy that uses water-based solutions to extract metals from ores, concentrates, and recycled materials. This process is widely used for recovering a variety of metals, including copper, gold, silver, zinc, nickel, cobalt, and lithium
Examples of Hydrometallurgy?
- Copper Extraction: Involves leaching copper with sulfuric acid, followed by solvent extraction and electrowinning.
- Gold Extraction: Utilizes cyanide leaching and the Merrill-Crowe process (zinc cementation).
- Recycling: Metals are recovered from electronic waste using hydrometallurgical techniques.
Key Steps in the Hydrometallurgical Process
Leaching: Dissolving the desired metal from its ore or material using an aqueous solution (leaching agent).
- Solution Purification: Removing impurities from the solution through methods like ion exchange, solvent extraction, or carbon adsorption.
- Metal Recovery: Extracting the pure metal from the purified solution via techniques such as electrowinning or precipitation.
- Refining (Optional): Further purifying the metal to achieve ultra-high purity if required.
What is Metal Leaching?
Metal leaching is the process of dissolving a specific metal from its ore or material by exposing it to a chemical solution. This selective dissolution allows for efficient extraction of valuable metals.
What is the Cementation Process in Hydrometallurgy?
Cementation is a method of metal recovery where a more reactive (electropositive) metal is added to a solution containing ions of a less reactive metal. The less reactive metal precipitates out as a solid while the more reactive metal dissolves into the solution.
How is Copper Extracted Using Hydrometallurgy?
Copper extraction typically involves these steps:
- Leaching copper-bearing materials with dilute sulfuric acid.
- Purifying the resulting solution using solvent extraction.
- Recovering pure copper through electrowinning.
Which Metals Can Be Extracted Using Hydrometallurgy?
Hydrometallurgy is used to extract a wide range of metals, including but not limited to:
- Precious metals like gold and silver.
- Base metals like copper, zinc, and nickel.
- Specialty metals like cobalt, lithium, and uranium.
This method is particularly effective for low-grade ores and complex materials that are challenging to process through other techniques.